DNP Launches Recyclable Mono-Material Medical Sterilization Pouch
Achieves seal strength and peel-open performance comparable to conventional medical sterilization packaging, while helping medical device manufacturers improve recyclability
April 16, 2026
Tokyo, April 16, 2026, Dai Nippon Printing Co., Ltd. (DNP) has started providing samples of the newly developed “DNP’s Mono-material Medical Sterilization Pouch”. The pouch complies with the guidelines*1 of the Circular Economy for Flexible Packaging (CEFLEX) consortium and features a mono-material design that improves sortability and recyclability in the recycling process.
Composed of more than 90 wt% *2 polyethylene-based materials, the product will support medical device manufacturers in their efforts to realize a resource-circulating society. In addition, by leveraging our proprietary converting (material processing) technologies, the new pouch achieves seal strength equivalent to conventional sterilization pouches while enabling peel-open functionality without generating lint.
 DNP’s mono-material medical sterilization pouch
DNP’s mono-material medical sterilization pouch
Background
In the European Union (EU), adoption of recyclable mono-material packaging is accelerating, as requirements related to resource circulation and recyclability for packaging materials are being strengthened, including the implementation of the Packaging and Packaging Waste Regulation (PPWR) in February 2025. Currently, some recycling requirements are waived in the medical field, but to realize a resource-circulating society in the future, some medical device manufacturers are beginning to investigate ways to reduce the environmental impact by utilizing mono-material structures. DNP has developed the new mono-material sterilization pouch in response to these needs.
Key Features
1. Packaging design combines a mono-material structure with seal strength and easy opening comparable to conventional products
DNP’s Medical Sterilization Pouch is composed of a nonwoven substrate and a film. This enables sterilization of medical devices while enclosed within the package and is used in medical settings.
By leveraging proprietary DNP converting technology, the mono-material facilitates pouch manufacturing with mono-material structures, which previously presented challenges such as heat resistance, while achieving easy opening comparable to conventional products. Composed of more than 90 wt% polyethylene-based materials, the product is designed to offer improved sortability in the recycling process, and compatibility in the resource recovery process compared to conventional composite materials.
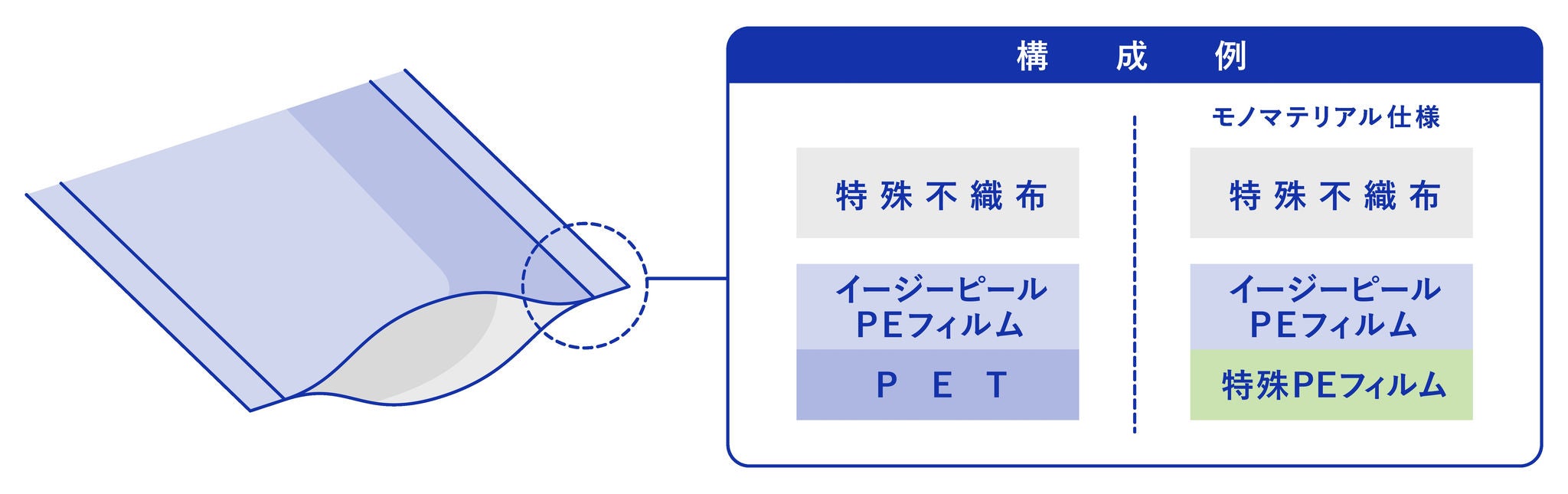 Comparison of conventional medical sterilization pouch and the mono-material type configurations.
Comparison of conventional medical sterilization pouch and the mono-material type configurations.
2. Compatible with a wide range of sterilization methods used by medical device manufacturers, and with a proven adoption track record
The new pouch is compatible with a variety of sterilization methods, including Ethylene oxide gas (EOG), Gamma radiation, and Hydrogen peroxide gas. DNP’s Medical Sterilization Pouch materials have a proven track record of adoption by a variety of medical device manufacturers and can also be customized into a variety of sizes and formats to meet specific application requirements.
Going Forward
DNP will supply the new pouch to medical device manufacturers and companies that process and convert sterilization packaging materials, aiming for cumulative sales of approximately JPY 400 million by FY 2030. In addition, DNP will exhibit the new product at Booth No. 1014 (East Hall 7) at Medtec Japan, a trade show for the design and manufacture of medical devices, to be held at Tokyo Big Sight from April 21st to 23rd, 2026.
*1 CEFLEX Guidelines: The Designing for a Circular Economy (D4ACE) international design guidelines for improving the recyclability of film or flexible packaging. These guidelines establish standards for material composition aimed at improving sortability and recycling quality.
*2 wt%: The ratio representing the weight of the component relative to the total weight.
*All company and product names mentioned herein are the trademarks or registered trademarks of their respective owners.
* The information contained in this release is current as of the date of announcement and is subject to change without notice.
※DNP医療用滅菌包材 モノマテリアルタイプに関するお問い合わせ → https://biz.mkt.global.dnp.co.jp/l/819623/2026-04-07/647zq7
