DNP and Hyperion Drug Discovery Develop Soluble Microcarriers used in Cell Culture for Regenerative Medicine
Improves production efficiency
Mar 16, 2023
Dai Nippon Printing Co., Ltd.
Hyperion Drug Discovery Co., Ltd.
Tokyo, March 16, Dai Nippon Printing Co., Ltd. (DNP) and Hyperion Drug Discovery Co., Ltd. (HDD), that develops regenerative medicine and gene therapy, have developed soluble microcarriers used as scaffolding agents in cell culture processes, such as cell and gene-based products, exosomes, biopharmaceuticals, and cultured meat.
Microcarriers are materials used for three-dimensional culture by attaching cells to their surface in a culture medium. Compared to conventional two-dimensional culture, such as petri dishes, this makes it possible to reduce staff levels in line with a cutback in the number of work processes. It is also possible to cut down the necessary installation space while facilitating efficient large-scale cell culture.
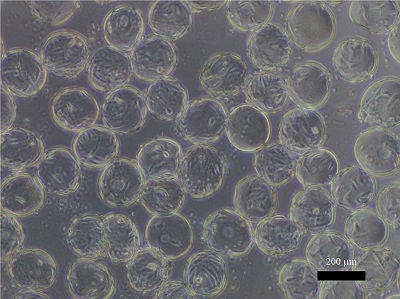
Microscopic image of human mesenchymal stem cells on the surface of microcarriers. (Provided by Kino-oka Laboratory, Graduate School of Engineering, Osaka University)
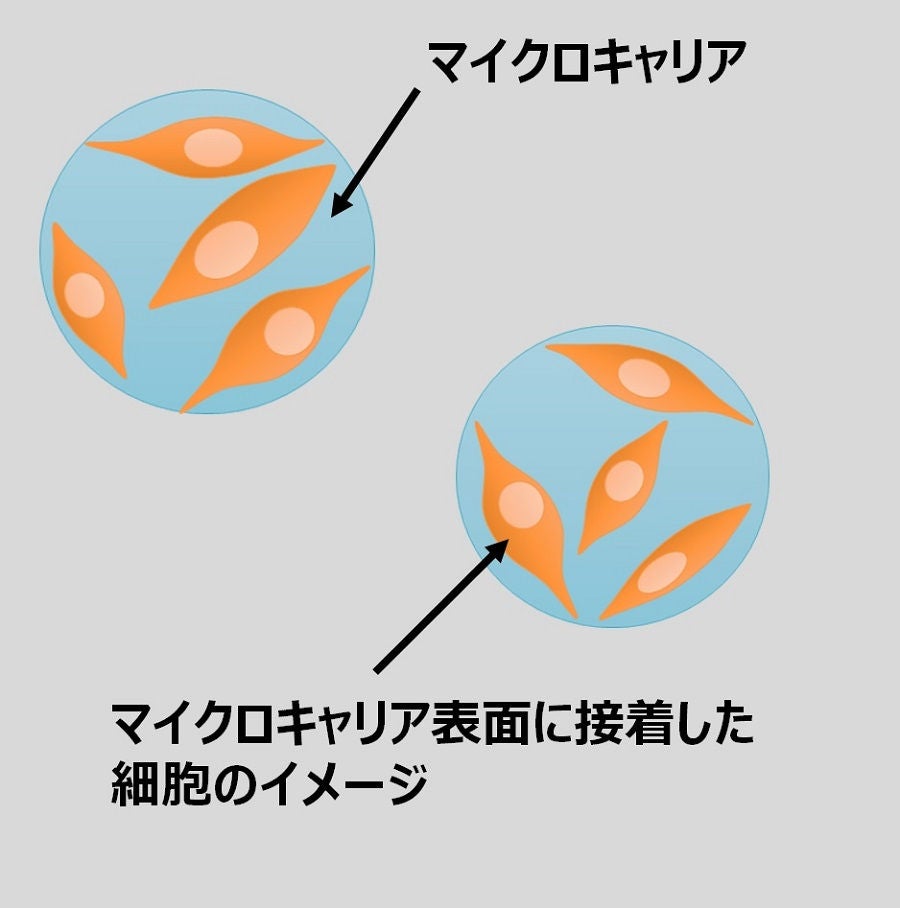
Image of microcarrier culture
Image of microcarrier culture
The cell culture-related market has recently undergone expansion in line with the growing needs of cell and gene-based products and biopharmaceuticals. Numerous cells are known to adhere peripheral environment and proliferate, so surface design suitable for cell culture has become important. In conventional methods, cells are cultured by modifying the surface of petri dishes, otherwise referred to as two-dimensional culture. Consequently, as the number of cells increases, the number of petri dishes also increases, requiring a large culture space. In addition, in line with similar work being repeated for each petri dish, there has also been the challenge of needing to increase the number of required personnel, as well as a rise in cases of human error. To address this issue, a culture method using the surface of microcarriers, or three-dimensional suspension culture, has begun to be used. However, many non-soluble polymer products are used as microcarrier materials, presenting the potential for microcarriers in culture colliding with each other resulting in the release of tiny fragments. There was also a risk that these fragments could not be completely removed with a filter, and may permeate into the formulation. In response to these challenges, the two partners have combined DNP's coating technology and know-how in material selection with HDD's cell culture technology applicable to clinical use and knowledge of compliance with pharmaceutical regulations to facilitate mass cell culture. As a result the partners have successfully developed a soluble microcarrier that enhances safety to comply with pharmaceutical regulations.
Soluble Microcarrier Features
Achieving work efficiency and cost reductions via larger scale culture based on space savings
Compared to the current mainstream flat culture on a petri dish, the newly developed microcarrier reduces staff levels in line with a cutback in the number of work processes, and the overall manufacturing process. It also improves production efficiency by reducing culture space and improving manufacturing efficiency. For example, according to HDD, the new microcarrier can reduce the culture space and the amount of culture medium used by 75%. In cell-based products, it is sometimes necessary to administer more than 100 million cells per patient. As a result, the new microcarrier is not only effective for mass culture, but cost reductions can also be expected.
Highly Safe Materials reduce Damage to Cultured Cells
The main material of the new microcarrier is a highly biocompatible algae-derived sodium alginate soluble gel. Unlike conventional insoluble polymer products, it is a safe product that does not generate insoluble microscopic fragments even when microcarriers collide with each other during culture. In addition, by utilizing a separation method different from insoluble polymer products, it is simple to recover only cultured cells by dissolving the microcarrier using a detachment agent commonly used in regenerative medicine. This results in less damage to cells and leads to improved work efficiency and quality.
Eligibility as Material for Regenerative Medical Products Confirmed by Regulatory Authority in Japan
The eligibility of the new microcarrier has been confirmed as a material for regenerative medical products by the Pharmaceuticals and Medical Devices Agency (PMDA) via a process of Consultation on the Eligibility of Materials for Regenerative Medical Products.
Going Forward
DNP and HDD have signed a joint development agreement on the efficiency of cell culture using microcarriers as a core technology. The partners will develop further technologies, along with products and services related to cell culture, contributing to the future development of regenerative medicine
DNP Initiative Related to Regenerative Medicine in the Medical Healthcare Field
DNP is focusing on developing new businesses in the medical and healthcare field by leveraging its strengths in its proprietary printing and information (P&I) business. In 2008, we were the first company in Japan to commercialize equipment that can culture cells in a stable manner, in various patterns, using thin-film multilayer patterning technology. We also manufacture temperature-responsive cultureware that makes it possible to neatly remove cultured cells from a sheet when the temperature is lowered.
About HDD
HDD was founded in 2020, and leverages strengths in responding to pharmaceutical regulations to promote the development of platforms, products, and peripheral technologies for regenerative medicine and gene therapy based on totally new mechanisms that have not existed to date.
Dai Nippon Printing Co., Ltd. Location: Shinjyuku-ku, Tokyo Company President: Mr. Yoshinari Kitajima
Hyperion Drug Discovery Co., Ltd. Location: Nara City, Nara Prefecture President/CEO: Mr. Kazuhiro Takekita
- Company and product names referred to in this release are the trademarks or registered trademarks of their respective owners.
- Product specifications and service details referred to in this news release are current as of the date of the announcement. They may be changed without notice.
